Fourier Transform Infra-Red
Theory of Operation
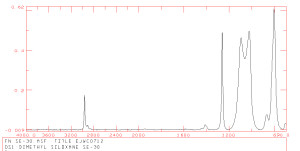
FTIR-1
The component atoms of polyatomic molecular groups are in constant dynamic state of motion with respect to each other. They are changing between the molecular ground state and quantum mechanically allowed excited states due to thermal excitation. This movement allows the spectroscopist to observe the twisting, bending, rotating and stretching motions of the atoms within a molecule occur at frequencies that are in the infra-red (IR) portion of the electromagnetic spectrum (0.7500 um). When infra-red light of energy coincident with the difference between the ground state and excited state of a particular molecular vibration is radiated onto a sample, the vibrations are stimulated (it jumps from the ground state to an excited state), and the light of that particular wavelength is absorbed by the molecule (IR absorbance). By monitoring the absorbance of the a range of wavelengths or frequencies of an infra-red light as it is transmitted through or reflected from (IR reflectance) a sample, a characteristic infra-red spectrum can be obtained (as above). In Fourier transform infra-red spectroscopy, the source is modulated with a Michelson interferometer. This results in a signal at the detector which has a distribution of frequencies determined by the speed of the moving mirror of the interferometer. The resulting interferogram in the amplitude time domain is then transformed via the Fourier algorithm into the appropriate amplitude-wavelength domain, resulting in a characteristic infra-red spectrum for a particular compound.
Typical Applications
- Identification of extractable organic components from a device or wafer surface or PWB
- Identification of contamination (oils, polymers, solvents, fluxes, greases, etc.)
- Identification of unknown solid organic compounds (powders or residues and solvent or liquid organic)
- FTIR is the leading analytical instrumentation for identification of organic substances
Sample Requirements
Because of the improved performance over former dispersion type IRs, much less sample is required for the analysis. Sample size, shape and physical state restrictions apply to all of the methods outlined above and require sample preparation. When performing an analyses on an electrical device, it is desirable to perform a side-by-side analysis of a “good” part and a “failed or rejected” part in order to distinguish the chemical, surface or physical sample features that differentiate them. One must distinguish what is “supposed to there” with " what is actually there" and the difference between the two is the unknown or contaminate.
As noted in the theory and applications sections describing the available instrumentation, many of the techniques are sensitive to elemental concentrations in the ppm to ppb range and/or to surface regimes on the order of 1-10 atomic layers. It is therefore imperative that samples to be analyzed are handled in an appropriate manner to avoid the introduction of extraneous contaminants prior to submission for analysis.